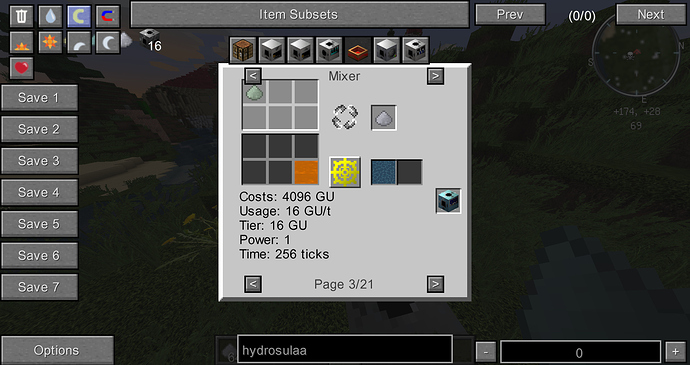
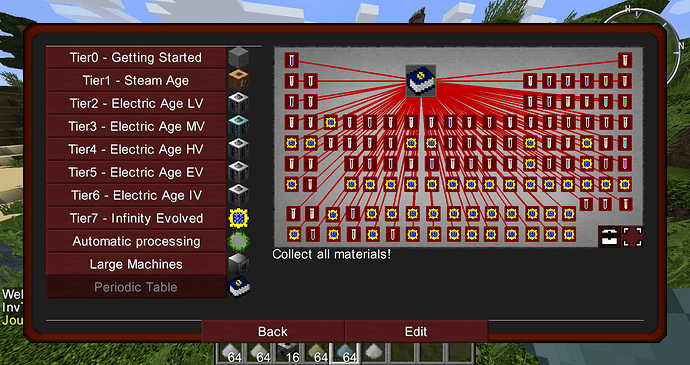
I found that some of the materials in GT6 are “useless”, they have no special purpose, nor are they ideal materials for tools. Some of them can be normally obtained, some can not be obtained, I think they have some possible uses…
Cerium: It can be obtained from bastnaesite or centrifugal rare earth. It is useless in GT5 and is not an ideal material for tools. It can make some special alloys and catalysts in reality, but GT6 does not seem to need these alloys…
Selenium: Byproducts of minerals such as cinnabar. Because GT printers don’t need selenium drums, I think it can be used to make higher tier solar panels…
Germanium: Byproducts of stannite and kesterite, GT6 does not seem to need germanium to participate in the more complex semiconductor industry… Maybe they can be used as photoelectric components and catalysts? (Germans should be able to do that)
Ruthenium and Rhodium: Maybe the best use of platinum mud or byproduct as an “expensive waste” is as a catalyst…
Sulfite: I don’t know what use it is… Maybe they can be used to restore? Sodium dithionite (commonly known as “insurance powder”) is a reductant, which can be used for bleaching and synthesis of thiourea, etc.
Pyrosulfate: They are not normally available, they are useless, and I don’t know why they are added… They seem to dissolve some metal oxides like persulfates.