Hi, @Gregorius, Recently, you have seen that you are adding multi-block structures, which facilitate large-scale production. I recall the suggestion that ammonia should be produced with a special multiblock structure, so I hope you can add ammonia in the next version. Ammonia is very important in the actual chemical production. They are mainly used to produce fertilizers and other chemical products. Although minecraft does not need too much fertilizer, ammonia can still produce many important chemical raw materials. I see ammonia now. There are no recipe and a lot of nitrogen produced by centrifuging air is useless. I hope to add ammonia to the synthesis.
In reality, ammonia is synthesized by reacting nitrogen and hydrogen under high temperature, high pressure and catalysts. The catalysts used in the reaction may differ somewhat in composition in different places, but the main components are Fe3O4 (although new catalysts have been developed, they are not widely used). The structure of the reactor is usually as follows: nitrogen and hydrogen enter from the top, then reach the bottom of the heat exchanger, heat exchange with the former reacted gas, and then enter the reaction chamber with electric heating wire and catalyst in the middle. The reacted gas is discharged from the bottom of the reactor after cooling through the heat exchanger, because only 10-20% of the reacted gas is ammonia generated after the reaction. In order to separate, the remaining nitrogen and hydrogen after separation are returned to the reactor for recycling (the game may not need to be so complicated… ) In the game, you may need steel to be able to withstand high temperature and high pressure to make a shell, put a reaction chamber in the middle, input electricity in the middle at the top, input raw gas at the top, and output reacted gas at the bottom.
What’s more, have you thought about the production method of hydrogen peroxide?
For NH4 I do not see many uses, that’s why its not added yet.
As for H2O2 on the thread where there was talk about a way to synthesize it, someone said there was a method, but I did not get any sources for that method whatsoever, so that got skipped because they did not even tell me how exactly that one worked at all.
In fact, ammonia is one of the most produced inorganic substances in the world. More than 80% of them are used in the production of fertilizers, and the rest are also used in a large number of ways. There are some reactions about ammonia in GT5, which can be used as a reference.
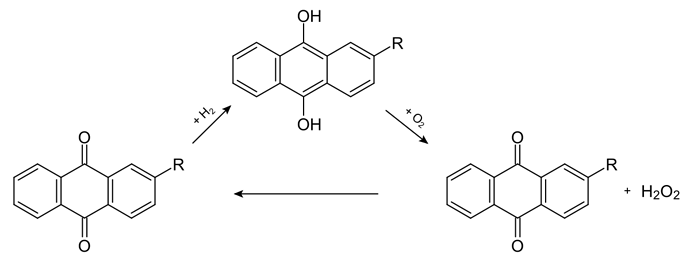
As for hydrogen peroxide, Wikipedia should have a description of its production process. You can check the relevant information to know how to produce it. Anthraquinone process is the mainstream production method nowadays.
I am not gonna look at GT5U, so tell me actual uses for Ammonia that aren’t Fertilizer.
Wikipedia DOESN’T have info on that particular process that got mentioned, it has info on other processes but not on the one that got mentioned.
The electrolytic preparation method of hydrogen peroxide is clear enough.
1.Turning ammonium hydrogen sulfate (NH4HSO4) to ammonium persulfate [(NH4)2S2O8],by electrolyze the 1:4 mixed solution of sulfuric acid (H2SO4) and ammonium hydrogen sulfate.
2NH4HSO4==(NH4)2S2O8+H2
2.After that, ammonium persulfate need to pour into water.
(NH4)2S2O8+2H2O==2NH4HSO4+H2O2
3.Distilling H2O2 out by vacuum distillation.The NH4HSO4 can be recycled.
There are many papers which talk about this method in China.I also found this reaction pathway in the university chemistry textbook.But I can not get that book immediately,so I will check it tomorrow,
In fact, most ammonia is used to produce ammonium nitrate, a substance that is both fertilizer and explosive. If it is not needed as fertilizer, it can be used as a cheaper explosive, commonly known as “fertilizer explosive”, to make some explosives. Moreover, ammonia is also needed for ammonium bisulfate (although I’m not sure whether sodium/potassium persulfate is OK), as @dnk1234567 said, in the production of hydrogen peroxide, so ammonia has a very broad application prospect.
Here are some GT5U chemical reactions related to ammonia:
nitrogen+hydrogen=ammonia
distill fermented biomass=ammonia
ammonia+oxygen=nitrogen monoixde+water
ammonia+oxygen=nitric acid+water(in large chemical reactor)
ammonia+hypochlororus acid=chloramine+hydrochloric acid
ammonia+methanol=dimethylamine
ammonia+methanol+hypochlororus acid=1,1-dimethylhydrazine+hydrochloric acid+water(in large chemical reactor)
That was exactly what I looked for on Wikipedia and didnt find. Thank you for showing where the Electrolysis is actually applied to.
In fact, in addition to electrolysis, anthraquinone is the broader method of producing hydrogen peroxide, but the production of 2-ethylanthraquinone is more troublesome, so it can be used as a method that does not need electricity but will be troublesome at first.
I have referenced five inorganic chemistry textbooks.Here are them:
[1]Zhao Fengyi, Guo Dewei, Gui Mingde.Inorganic Chemistry Series Volume 5[M].Beijing:Science Press,1990:92-93.
[2]Zhang Zude.Inorganic Chemistry[M].Hefei:China University of Science and Technology Press,2018.
[3]Song Tianyou, Xu Jianing.Inorganic Chemistry Volume II[M].Beijing:Higher Education Press,2015.
[4]N.N.Greenwood, A.Earnshaw.Elemental Chemistry(Middle Volume)[M].Beijing:Higher Education Press,1996.
[5]Yan Xuanshen, Wang Changfu.General Inorganic Chemistry[M].Beijing:Peking University Press,2016.
I checked the part of Oxygen and Sulfur,including persulfate and hydrogen peroxide.I found different book giving different reactions,but the method I gave, that is, the method of hydrolysis of (NH4)2S2O8, is the majority.
(In fact,the literature [4] denied this method,[1] gave the Ammonium salt-Potassium salt combination method,and [3] only gave the method of K2S2O8.)
There are also some papers:
[1]Zhao Ke, Li Shuxian. Production method and application of hydrogen peroxide[J]. Chlor-Alkali Industry, 2000, 11: 22-23.
[2]Berglin.Method in the production of hydrogen peroxide[P].US4552748,1985.
In summary,although there is no such method on Wikipedia,the electrolysis method is truly exist and had been used.
(I think this may happen because of the difference in the region.For example,in sulfuric acid industry,the preparation of SO2, sulfur is used in European countries, but pyrite (FeS2) is used in China.That makes difference in books and documents. )
)
I just saw a more general way to produce nitric oxide than GT6 in reality… Nitric oxide and water are produced by ammonia and oxygen at high temperatures using platinum-rhodium alloy catalysts. There is this recipe in the chemical reactor of GT5. I don’t know what machine is used in GT6 to process this recipe. It’s more reasonable than GT6 is now.